Engineering Precision In Chromatography From Development To Commercial Scale
24 April 2026 | Friday | Analysis

Chromatography in Bioprocessing
For pilot and commercial purification of monoclonal antibodies and recombinant proteins, chromatography remains the central downstream separation technology because it can deliver orthogonal selectivity against process- and product-related impurities while still fitting GMP manufacturing logic. What has changed is the optimisation target. In modern bioprocessing, the problem is no longer simply “how do we reach purity?”, but “how do we reach purity reproducibly, at plant scale, with acceptable resin utilisation, buffer consumption, cycle time, cleaning burden, and cost of goods?”. That shift is why affinity capture, ion exchange polishing, membrane adsorbers, mixed-mode resins, and model-based development are now discussed in the same breath rather than as separate topics.
First, each chromatographic mode solves a different mechanistic problem: affinity gives the highest selectivity, ion exchange resolves charge-driven impurity differences, hydrophobic interaction exploits surface hydrophobicity, size exclusion separates by hydrodynamic size but suffers in preparative productivity, and mixed-mode ligands widen the design space by combining several interactions in one medium. Second, resin performance is inseparable from matrix chemistry and hardware. Agarose, cellulose, polyvinylether, polymethacrylate, polymeric membranes, and monoliths differ materially in pore architecture, rigidity, pressure-flow behaviour, alkaline tolerance, and mass-transfer regime. Third, scale-up is an engineering exercise, not a linear extrapolation from a bench column: residence time, bed height, dead volume, pressure drop, packing quality, and extra-column effects can materially change peak shape and capacity. Fourth, the regulatory endpoint is lifecycle control. Both the U.S. Food and Drug Administration[2] and the European Medicines Agency expect process understanding, validation, and continued verification, while PAT is increasingly used to support pooling, monitoring, and potentially real-time decision-making.
The strongest contemporary angle is future-oriented but not speculative. Continuous multi-column capture improves resin utilisation and throughput; single-use and pre-packed formats reduce packing and cleaning friction; and AI/ML is already becoming useful for surrogate modelling, soft sensing, and faster design-space exploration, especially when combined with mechanistic models and digital twins. The near-term opportunity is therefore not “fully autonomous purification”, but better hybrid decision support across development, scale-up, and routine manufacturing.
Scope and assumptions
In en-GB, with two target molecule classes: monoclonal antibodies and recombinant proteins. It also assumes pilot and commercial facility contexts rather than discovery-only purification. For Fc-containing antibodies, the baseline process architecture is affinity-led capture followed by orthogonal polishing; for recombinant proteins without Fc, the article should discuss affinity-tag capture where relevant but emphasise the same downstream logic of orthogonality, impurity clearance, scale-up discipline, and control strategy. Those assumptions are consistent with current bioprocess development practice and with the way therapeutic protein purification is treated in the literature.
Fundamentals of chromatographic methods and media chemistries
Affinity chromatography is the most selective mode because the stationary phase carries a biologically or structurally recognisable ligand. In antibody processing that usually means Protein A for Fc capture, but affinity can also include Protein L, CH1-selective media, and tag- or motif-based ligands for recombinant proteins or more complex antibody formats. Its great advantage is step compression: a well-designed affinity capture can operate directly on clarified harvest and can remove large fractions of host-cell proteins and DNA in one step. Its main liabilities are media cost, ligand leaching, and sensitivity of lifetime to cleaning chemistry and fouling.
Ion exchange chromatography should be presented as the workhorse of polishing and, in some cases, intermediate purification. Cation exchange binds species that are net positive under the chosen conditions; anion exchange binds net negative species. In practice, both bind-elute and flow-through modes are used. For monoclonal antibodies, cation exchange is often tuned to resolve aggregates and charge variants, while anion exchange is frequently used in flow-through mode to clear DNA, host-cell proteins, endotoxin, and sometimes viruses when the product remains largely unbound. The technical article should emphasise that ion exchange is rarely “generic”: performance depends strongly on pH, conductivity, gradient structure, ligand density, and the electrostatic heterogeneity of both product and impurity pools.
Hydrophobic interaction chromatography should be framed as an orthogonal complement to ion exchange. Proteins are loaded at elevated salt, which promotes interaction between hydrophobic surface patches and the ligand, then eluted as salt strength falls. HIC is valuable when charge-based selectivity is insufficient, including for aggregate or variant control in some antibody and recombinant protein processes. Its weakness is not conceptual but operational: it usually requires salt conditioning and therefore adds buffer burden and integration complexity. That is why HIC remains important, but often less universal than ion exchange in platform mAb processes.
Size exclusion chromatography should be described honestly. It is elegant, mild, and genuinely useful, especially for aggregate analysis, desalting, buffer exchange, and limited polishing of sensitive proteins; molecules elute according to hydrodynamic size because large species access less pore volume and therefore emerge first. But classical SEC remains handicapped in preparative bioprocessing by low loading capacity, slow operation, and weak scalability relative to other modes. That is why SEC is foundational analytically and still relevant technically, yet often not the economic centrepiece of large-scale therapeutic protein purification.
Mixed-mode chromatography now deserves more prominence than many older reviews gave it. Mixed-mode ligands deliberately combine two or more interaction types, commonly ionic with hydrophobic, aromatic, and hydrogen-bonding contributions. That expanded interaction space can make impurity clearance more robust, can improve aggregate or fragment removal, and can sometimes replace a weaker conventional polishing step. For antibody processes, mixed-mode materials are increasingly discussed not as exotic tools, but as practical options when standard CEX/AEX/HIC combinations do not open a sufficient design space.
The matrix chemistry behind these modes is just as important as the ligand. Hydrophilic agarose supports remain attractive for large biomolecules because of their open pore structure and biocompatibility, whereas more rigid synthetic supports such as polyvinylether, polymethacrylate, and other polymeric matrices offer better high-flow and pressure performance. For example, Merck’s Eshmuno A is built on a rigid, hydrophilic polyvinylether matrix; Thermo Fisher’s POROS affinity media are promoted on rigid polymeric supports for high-throughput, high-flow capture; and Purolite’s Praesto CH1 uses an engineered affinity ligand on highly crosslinked agarose. These are not trivial material choices: they determine dynamic binding capacity at practical residence times, pressure drop at production flow rates, and compatibility with alkaline CIP.
Resin columns remain the dominant incumbent in bioprocess chromatography, but membranes and monoliths are growing because their through-pores enable largely convective transport, lower diffusional limitations, and short cycle times for large biomolecules. This makes them especially attractive for flow-through polishing, virus and DNA clearance, large-particle capture, and single-use deployment. The engineering trade-off is that classical packed beds still often win on binding capacity for smaller proteins and on fully mature industrial familiarity, while membranes and monoliths win on speed, simplicity, and large-species accessibility.
Column design, scale-up, and process development
For packed beds, the key variables are bed height, column diameter, aspect ratio, pack quality, frit and distributor design, compression strategy, system dead volume, pressure limit, and targeted residence time. The usual scale-up heuristic is to preserve a comparable residence time and loading regime while increasing diameter, but this should never be presented as a law of nature. It is a starting point that must be checked against pressure-flow curves, resin compressibility, and peak broadening. A recent study on pre-packed preparative columns found that extra-column broadening can be more than 50% of total band broadening for larger-volume columns, showing that tubing, adapters, and hardware become scale variables in their own right.
The analytical article should therefore explain scale-up as a decision tree. If pressure is not limiting and mass transfer is stable, increasing column diameter while keeping similar hydraulic conditions is often acceptable. If pressure or cycle time becomes limiting, the options include reducing bed height, choosing a more rigid matrix, switching to a membrane or monolith, or moving to multi-column operation. This is especially relevant in polishing, where recent work has shown that residence time and bed height can materially affect recovery and clearance in multimodal operations. In other words, “same chemistry, bigger column” is rarely enough as a scale-up philosophy.
Process development should be presented as a QbD-informed workflow rather than a series of empirical resin screens. The modern sequence is: define target product profile and critical quality attributes; characterise feed and impurity burden; shortlist chromatographic modes and media; use high-throughput screening, mini-columns, membrane devices, and automation to identify operating windows; fit mechanistic or hybrid models where useful; then confirm the process in pilot and engineering runs before process qualification. Miniaturisation and parallelisation are now standard because they allow faster, cheaper definition of operating windows and more robust purification design than purely empirical bench-to-pilot extrapolation. Increasingly, digital-twin approaches and model-enhanced design-space exploration are reducing the experimental burden further.
- a platform downstream process flow for mAbs and recombinant proteins, showing where each chromatographic mode normally sits;
- a matrix-and-ligand schematic comparing agarose beads, rigid synthetic beads, membranes, and monoliths;
- a scale-up decision tree linking residence time, pressure limit, dead volume, and format choice;
- a cost-driver graphic linking resin lifetime, cycle count, buffer load, and facility fit.
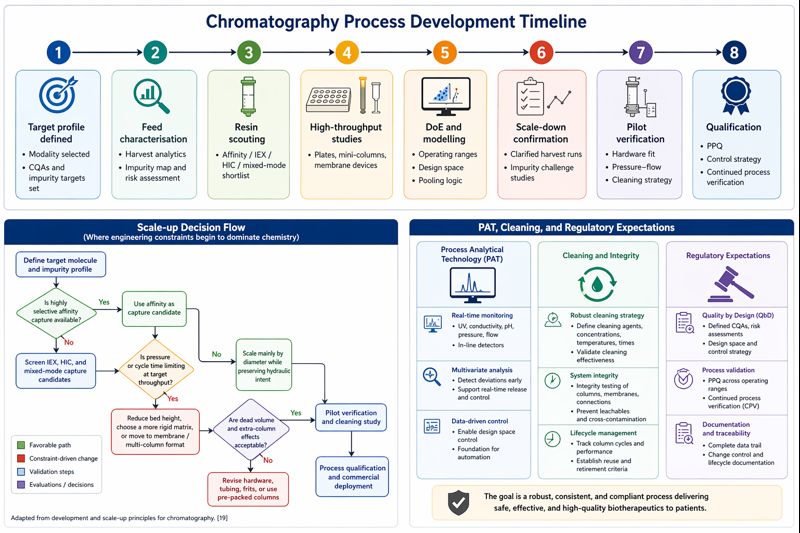
PAT, cleaning, and regulatory expectations
The PAT section should be explicitly linked to regulator thinking. The FDA’s PAT framework was written to encourage innovative pharmaceutical development, manufacturing, and quality assurance, and that logic maps well onto downstream chromatography. At minimum, chromatographic PAT includes UV, conductivity, pH, pressure, and flow trending; at the more advanced end it includes online HPLC, Raman spectroscopy, MALS, refractive index, ATR-FTIR, fluorescence, and sensor fusion models for pooling or concentration prediction. The practical message is that PAT in chromatography is moving from passive monitoring toward active decision support. Several studies have already demonstrated online HPLC for real-time pooling decisions, inline Raman with low-dead-volume flow cells, and multisensor monitoring for predicting purity and quantity during chromatographic capture. In protein A load monitoring, one comparative study found UV alone gave the most accurate concentration prediction, which is a useful reminder that “more sensors” is not automatically better unless the model and use case justify them.
Cleaning and sanitisation should be treated as central engineering and economics topics, not housekeeping. Sodium hydroxide remains the dominant CIP and sanitising agent because it is effective against precipitated proteins, hydrophobic foulants, nucleic acids, endotoxins, and many microorganisms and viruses, and because it is relatively cheap and straightforward to handle. Typical protein A sanitisation windows remain in the 0.1–0.5 M NaOH range, but the correct condition is media- and fouling-specific, and aggressive alkaline cleaning can shorten ligand lifetime. That is why resin suppliers now differentiate strongly on alkaline stability. Examples include Thermo Fisher’s alkaline-stable MabCapture C, Merck’s Eshmuno A claim of acid- and alkali-compatible cleaning while maintaining binding capacity, and Tosoh’s report that TOYOPEARL AF-rProtein A HC-650F retains 80% of initial dynamic binding capacity after 40 CIP cycles with 0.5 M NaOH. Membrane formats sometimes shift the problem entirely by being validated primarily for single use, thereby reducing cleaning-validation burden even if reuse is technically possible.
For regulation, the article should focus on lifecycle control rather than a checklist of guidances. The EMA process-validation guideline for biotechnology-derived active substances states that process characterisation and verification studies should normally be completed for the regulatory submission, and specifically requires evidence that downstream purification can both deliver the desired product and remove process- and product-related impurities. FDA process validation is similarly lifecycle-based, spanning process design, process qualification, and continued process verification. Under ICH Q8 and Q11, developers are expected to connect process understanding to design space, control strategy, and CTD Module 3 documentation. For biosimilars or major process changes, the same logic becomes even more demanding because impurity profiles, variant distributions, and process comparability must be justified with high analytical confidence.
Supplier landscape and case studies
The supplier comparison should make clear that “chromatography supplier” does not always mean the same thing: some firms are strongest in base media and ligands, some in membrane adsorbers, and some in pre-packed column platforms. That distinction matters because plant bottlenecks are heterogeneous. One site may be capacity-limited by affinity resin cost; another by polishing buffer use; another by packing and cleaning turnaround; another by the need to produce many molecules in smaller campaigns.
|
Vendor |
Product types |
Key features |
Typical applications |
Scale |
|
Sartorius |
Sartobind membrane adsorbers |
Cross-linked cellulose support with covalently attached ion-exchange ligands; primarily validated for single use; suited to flow-through impurity removal and large biomolecules |
DNA/HCP/endotoxin/virus clearance, aggregate reduction, some large-protein and viral-vector capture |
Multiwell to jumbo 5 L capsules/cassettes |
|
Thermo Fisher Scientific |
POROS affinity/IEX media; MabCapture C; CaptureSelect matrices |
Rigid polymeric supports for high-flow capture; alkaline-stable recombinant Protein A; domain-specific affinity such as CH1 |
mAb capture, Fab and bsAb capture, high-throughput process columns |
Process-scale packed columns from development to commercial manufacture |
|
Merck KGaA |
Eshmuno A, Eshmuno CPX, Fractogel |
Rigid hydrophilic polyvinylether matrix for Protein A; acid/alkali-cleanable; proprietary tentacle chemistry in CPX for high aggregate-removal efficiency |
Fc-protein capture, CEX polishing, aggregate clearance |
Pilot and commercial packed-bed operations |
|
Tosoh Bioscience[30] |
TOYOPEARL affinity/IEX/HIC media; TSKgel SEC |
Rigid, alkaline-resistant Protein A media; high DBC at practical residence times; documented NaOH CIP durability |
mAb capture, orthogonal polishing, SEC-based aggregate work |
Pilot to commercial packed-bed processing |
|
Bio-Rad Laboratories |
Nuvia cPrime, Nuvia aPrime, Nuvia ion exchangers |
Porous polymeric base matrices, low backpressure at high flow, mixed-mode selectivity with broad design space |
Mixed-mode polishing, AEX purification, therapeutic protein processing |
Lab-to-process scale |
|
Purolite |
Praesto affinity, CH1, IEX and HIC media |
Engineered affinity ligands on highly crosslinked agarose; high binding capacity and flow capability; NaOH-stability claims in affinity formats |
mAb/Fab capture, CH1-based bsAb purification, orthogonal polishing |
Process and commercial packed-bed use |
|
Pall Corporation |
Mustang membrane adsorbers |
PES-based membranes with quaternary amine functionality; constant bed depth across scales; high capacity for large biomolecules |
Impurity removal or capture from screening to manufacturing, especially DNA/virus/large-biomolecule duties |
Approx. 1 mL to 5 L |
|
Repligen |
OPUS pre-packed columns |
GMP-ready pre-packed columns with linear scale-up; intended to reduce packing equipment and turnaround burden |
Pilot, validation, and commercial campaigns using selected third-party media |
Sizes extending to large columns suitable for 1000–2000 L single-use bioreactor contexts |
The first is step elimination: Ichihara and co-workers reported an all-flow-through platform using activated carbon plus flow-through cation- and anion-exchange steps for therapeutic monoclonal antibodies, achieving excellent impurity clearance with overall yield above 80% at high loadings. The second is resin-utilisation intensification: a hybrid continuous mAb purification study reported comparable product quality to batch processing, but with protein A productivity increased by up to 420%, buffer use reduced by 30–40%, and at least 48 hours of robust continuous run time. The third is molecule-specific affinity diversification: recent work on CH1-specific affinity shows that such resins can separate heterodimer from homodimer impurities in asymmetric bispecific antibody purification, demonstrating that the future of “affinity” is broader than generic Fc capture. [40]
Taken together, those examples illustrate the three highest-value innovation levers in bioprocess chromatography: remove a step entirely, use existing resin more efficiently, or change the selectivity basis so that the impurity problem becomes easier. That is a more useful analytical framework than simply ranking media by binding capacity. [41]
Economics and future directions
The economics section should be explicit that chromatography is both a technical and a financial bottleneck. Column volume is driven by feed mass per campaign, effective dynamic binding capacity at the chosen residence time, target cycle time, and allowed reuse count. Cost then follows from resin/media price, lifetime, buffer generation and storage, labour, packing, cleaning validation, equipment occupancy, and campaign scheduling. For mAb platforms, Protein A remains especially important economically because affinity media are expensive and lifetime-limited relative to many polishing media; continuous capture and better alkaline stability are therefore significant not because they are fashionable, but because they directly alter the most expensive step.
At high utilisation, reusable packed beds can still be compelling because the resin lifetime is fully consumed. At lower utilisation, smaller multi-product facilities may rationally prefer pre-packed or single-use approaches, because reduced packing labour, reduced cleaning validation, and faster turnaround can outweigh the higher cost of consumables. That conclusion is an inference from the available technical and vendor evidence, but it is the economically important one for real facilities.
Future trends are converging around three themes. Continuous chromatography is now technically mature enough to be discussed as an implementation choice, not merely a research topic: PCC and related multi-column systems improve capacity utilisation and can cut buffer demand while raising throughput. Single-use and ready-to-deploy chromatography are expanding through membrane adsorbers and large pre-packed columns, reducing the friction between development and manufacturing. AI/ML and digital twins are becoming practical in process development and control, especially for surrogate modelling of chromatographic elution, data fusion from PAT sensors, and accelerated design-space assessment. The most credible forecast is therefore a hybrid future in which packed beds remain dominant for many core duties, while continuous operation, single-use deployment, and model-assisted decision-making progressively remove the classical bottlenecks of resin underutilisation, cleaning burden, and slow development cycles.
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks




