Optellum Secures TGA Approval In Australia For AI Powered Virtual Nodule Clinic
02 March 2026 | Monday | News
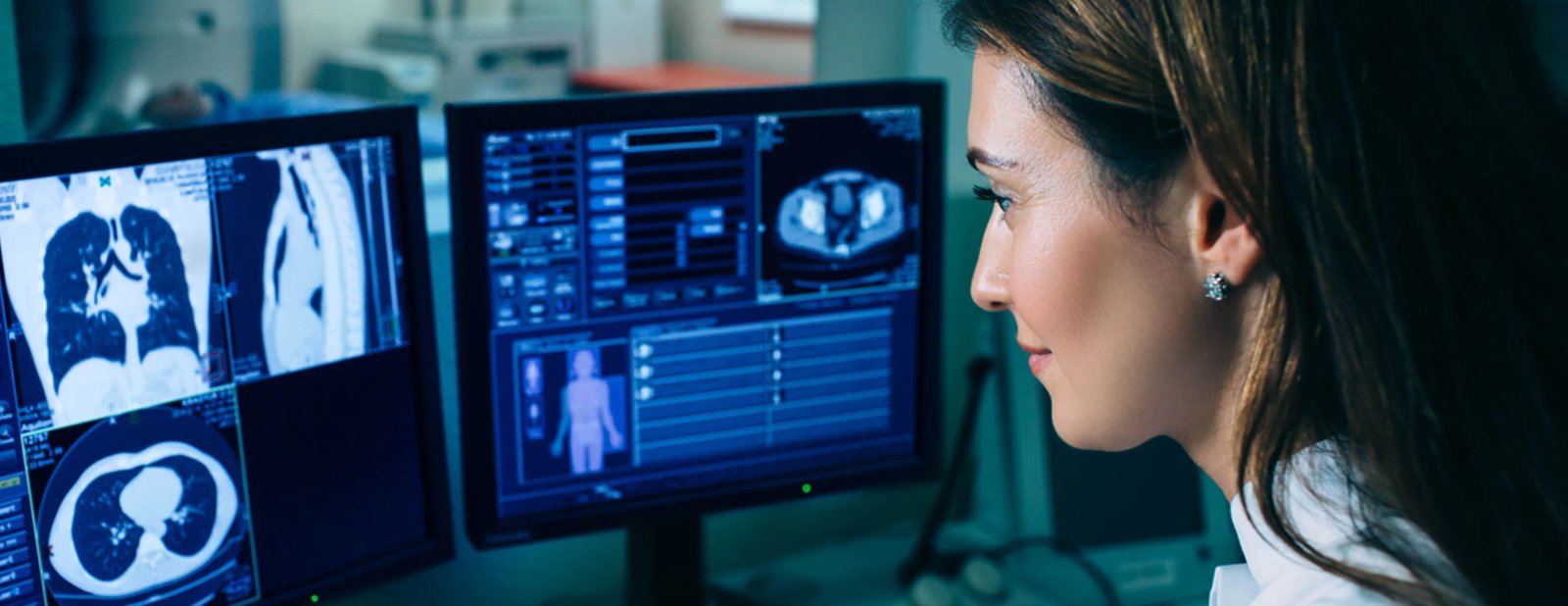
Optellum, the leading AI-enabled solution for earlier lung cancer detection, is thrilled to announce that it has received Therapeutic Goods Administration (TGA) class IIb approval in Australia for the Virtual Nodule Clinic (VNC) platform to support early lung cancer diagnosis.
This important regulatory milestone was achieved after a rigorous review of technical documentation and validation studies, recognizing its safety and performance for clinical use. VNC helps identify, triage, and manage patients with pulmonary nodules, and includes a clinically validated AI decision support tool, lung cancer prediction (LCP) AI, already used in the US and EU/UK for nodule risk stratification.
Lung cancer remains a major health challenge in Australia, with over 15,000 new diagnoses each year and low 5-year survival rates around 27%. [1] Delayed detection of incidental findings, inconsistent follow-up, and growing pressure on imaging and specialist services continue to contribute to later diagnosis.
These pain points can lead to unnecessary anxiety for patients and missed opportunities for earlier intervention, highlighting the need for tools that support timely recognition and coordinated health systems.
This TGA clearance poises Optellum to help thousands of patients in Australia by supporting earlier and more consistent identification of incidental lung nodules. By helping care teams prioritize higher-risk findings and streamline follow-up more efficiently, VNC aims to reduce delays and uncertainty along the lung cancer care pathway. This clearance marks an important step in bringing validated AI tools into everyday clinical workflows, without replacing clinical judgment.
Optellum remains committed to working with Australian health systems to support responsible AI adoption that improves patient care quality and experience. This milestone reflects Optellum's broader mission to help patients move from detection to diagnosis with speed, greater clarity, and confidence.
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks




