AGC announces strategic expansion of its Biopharmaceutical CDMO capability in Yokohama, Japan, signaling a new era of innovation and growth
26 December 2023 | Tuesday | News

This expansion has been selected by the Japanese Ministry of Economy, Trade and Industry (METI) as part of its "Developing biopharmaceutical manufacturing sites to strengthen vaccine production project" (announced by AGC in October 2022). AGC will introduce dual-use facilities that can be applied to manufacturing of vaccines in the event of a pandemic. In addition to mammalian cell culture bioreactors which have one of the largest*2 capacity as a CDMO in Japan, the expansion will also include facilities for leading-edge field of mRNA pharmaceuticals and gene and cell therapies. This will greatly contribute to the domestic development and manufacturing capabilities for biopharmaceuticals, which are currently highly reliant on overseas CDMOs. Furthermore, fully operational, it is expected to employ approximately 400 employees and contribute to the advancement of the society and biotech industry.
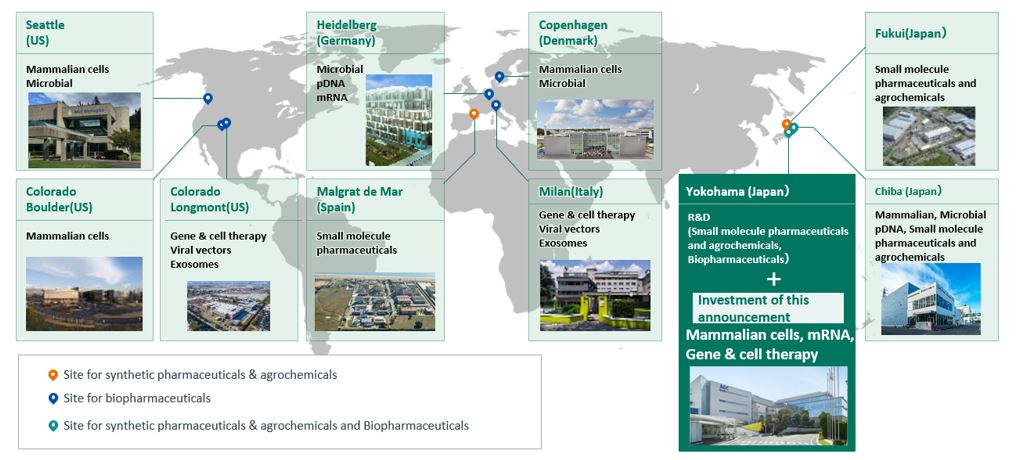
Under its medium-term business plan, AGC plus-2023, the AGC Group has positioned its Life Science business including its biopharmaceutical CDMO, as one of its strategic businesses. The Group is expanding its business through proactive acquisitions and capital investments. Until now, AGC has established cGMP*3-compliant service systems in Japan, the U.S., and Europe (see "Reference" below) and has built up an extensive track record. By combining the state-of-the-art technology and knowledge and experiences at these existing sites, AGC will swiftly establish a high-level service system at the Yokohama Site to meet the strong demand for biopharmaceutical development and manufacturing services not only in Asia including Japan, but also from all over the world.
Reference
1. The AGC Group's CDMO business sites and supported modalities*4
2. Overview of the investment
|
Summary |
|
|---|---|
|
Location |
Tsurumi-ku, Yokohama (AGC Yokohama Technical Center) |
|
Features |
- One of the largest capacity of bioreactors for mammalian cell cultures as a CDMO in Japan (AGC estimates). - Dual-use facilities that can be applied to vaccine production in the event of a pandemic. - Establishing a high-level service system by utilizing the state-of-the-art knowledge and experiences at existing sites in Japan, the U.S., and Europe. |
|
Service starting dates |
Existing facilities: 2025 (Development services for gene and cell therapies will start in advance) New facilities: 2026 (Start of development and manufacturing services for mRNA pharmaceuticals, biopharmaceuticals made using mammalian cell cultures, and gene and cell therapies) |
|
Total investment (estimated) |
Approximately 50 billion yen |
|
Number of employees (projected) |
When in full operation, facility expects to employ approximately 400 employees |
Notes
*1 CDMO: Contract Development & Manufacturing Organization. A company which is contracted on behalf of another company to provide manufacturing services of pharmaceuticals, etc. as well as the development of manufacturing processes.
*2 AGC estimates.
*3 Standards for manufacturing and quality control of the latest pharmaceuticals and quasi-drugs
*4 Terms that describe the classification of methods and means of basic technologies for drug discovery (e.g., mRNA pharmaceuticals).
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks




