Takeda Reports Strong Phase 3 Results For Zasocitinib In Plaque Psoriasis
30 March 2026 | Monday | News
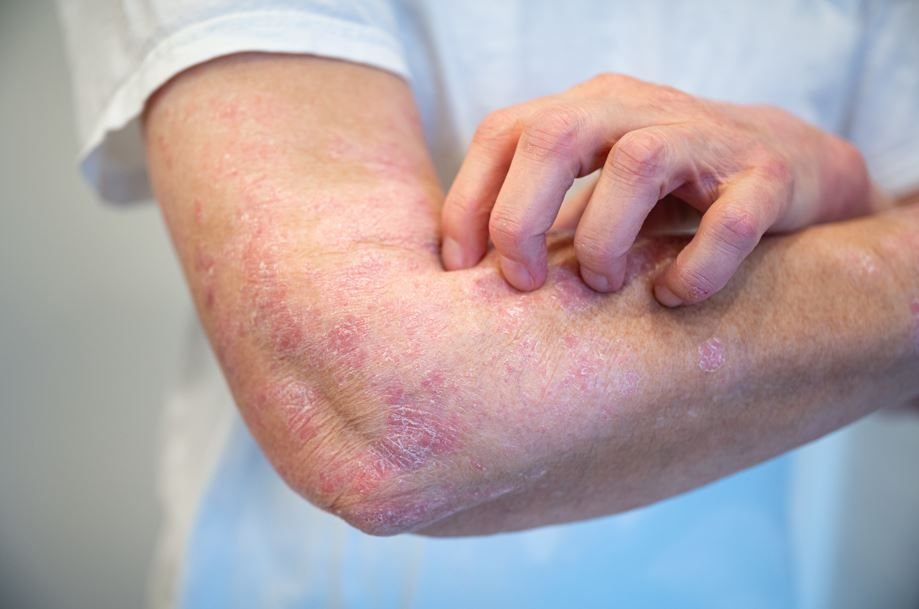
- About 70% of patients treated with zasocitinib achieved clear or almost clear skin (sPGA 0/1) at week 16 in Phase 3 plaque psoriasis studies
- A significantly greater PASI 75 response rate versus placebo was observed as early as week 4
- Safety profile consistent with Phase 2b studies with no new safety signals identified
Takeda announced new data from the two pivotal Phase 3 studies of zasocitinib (TAK-279), a next-generation, highly selective oral tyrosine kinase 2 (TYK2) inhibitor, in adults with moderate-to-severe plaque psoriasis (PsO).1 Presented as a late-breaking abstract at the 2026 American Academy of Dermatology (AAD) Annual Meeting, these data show that convenient once-daily oral zasocitinib demonstrated rapid and durable skin clearance with a safety profile consistent with Phase 2b studies.1,2
“These efficacy and safety results show it’s possible for a once-daily pill to deliver rapid, lasting skin clearance, highlighting the potential of zasocitinib to become a leading oral treatment option for plaque psoriasis.”
“Our goal in psoriasis treatment is clear or almost clear skin, and previously this has been achieved primarily with injectable therapies,” said Melinda Gooderham, MSc, MD, FRCPC, dermatologist, SKiN Centre for Dermatology, Peterborough, Ontario, Canada, principal investigator for the Latitude PsO studies and presenting author. “These efficacy and safety results show it’s possible for a once-daily pill to deliver rapid, lasting skin clearance, highlighting the potential of zasocitinib to become a leading oral treatment option for plaque psoriasis.”
In the Phase 3 randomized, multicenter, double-blind, placebo- and active comparator-controlled Latitude PsO 3001 and 3002 studies, more than half of patients treated with zasocitinib achieved clear or almost clear skin at week 16, a key measure of treatment success: 1,2
- 71.4% and 69.2% of patients treated with zasocitinib achieved a static Physician Global Assessment (sPGA) score of 0/1 versus placebo (10.7% and 12.6%) and apremilast (32.1% and 29.7%) at week 16 (p<0.001).2
- 61.3% and 51.9% of patients treated with zasocitinib achieved Psoriasis Area and Severity Index (PASI) 90 versus placebo (5.0% and 4.0%) and apremilast (16.8% and 15.9%) at week 16 (p<0.001).2
Zasocitinib also demonstrated statistically significant improvements in complete skin clearance, an increasingly important treatment goal for patients with plaque psoriasis:1,2
- 39.9% and 33.7% of patients treated with zasocitinib achieved an sPGA score of 0 versus placebo (0.7% and 1.4%) and apremilast (8.0% and 6.5%) at week 16 (p<0.001).2
- 33.4% and 25.2% of patients treated with zasocitinib achieved a PASI 100 versus placebo (0.7% and 1.1%) and apremilast (2.9% and 4.3%) at week 16 (p<0.001).2
- Responses for co-primary and key secondary endpoints continued to increase through week 24 in both studies.2
In Latitude PsO 3002, rapidity of response was demonstrated as early as week 4 versus placebo (PASI 75: 16.8% for zasocitinib vs 4.3% for placebo, p<0.001).2 Among patients who achieved a PASI 75, PASI 90 or sPGA 0/1 response at week 40 and continued on zasocitinib throughout the study, over 90% maintained their response at week 60.2
Zasocitinib was generally well-tolerated.1,2 The safety and tolerability profile of zasocitinib in the Phase 3 studies remained consistent with prior studies. 1,2 Key findings across the two studies include:
- Treatment-emergent adverse events (TEAEs) through week 16 were 62.1% for zasocitinib, 46.9% for placebo and 50.5% for apremilast.2
- The most common adverse events for zasocitinib treated patients through week 16 (≥5%) were upper respiratory tract infection (10.1%), nasopharyngitis (6.2%) and acne (6.5%), with no new safety signals identified.2
- Serious TEAEs through week 16 were 3.0% for zasocitinib, <1% for placebo and 1.5% for apremilast.2
“Our Phase 3 results demonstrate that highly selective TYK2 inhibition can offer many people with moderate-to-severe plaque psoriasis the potential for clear or nearly clear skin,” said Chinwe Ukomadu, MD, PhD, senior vice president and head, Gastrointestinal & Inflammation Therapeutic Area Unit at Takeda. “The positive data also underscore zasocitinib’s potential to deliver rapid and durable results with a favorable safety profile consistent with Phase 2b studies. We are working as quickly as possible with regulators to advance a potential new therapeutic option for patients seeking a safe, effective and convenient oral treatment.”
Takeda is on track to submit a New Drug Application with the United States Food and Drug Administration and other regulatory authorities starting in fiscal year 2026.
Results from the Phase 3 studies have no significant impact on the full-year consolidated forecast for the fiscal year ending March 31, 2026.
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks




