BioPharma Drug Approval
RemeGen’s Telitacicept Secures EMA Orphan Drug Designation for Myasthenia Gravis, Advancing Global Development
RemeGen Co., Ltd. ("RemeGen", stock symbols: 688331.SH/09995.HK) announced that telitacicept (RC18; brand name: 泰爱®) has received Orphan Drug Desig...
June 18, 2025 | News
Harrow Secures Exclusive U.S. Rights to BYQLOVI for Post-Surgical Eye Care Following FDA Approval
Harrow (Nasdaq: HROW), a leading North American eyecare pharmaceutical company, and Taiwan-based Formosa Pharmaceuticals ("Formosa", 6838.TW), today a...
June 10, 2025 | News
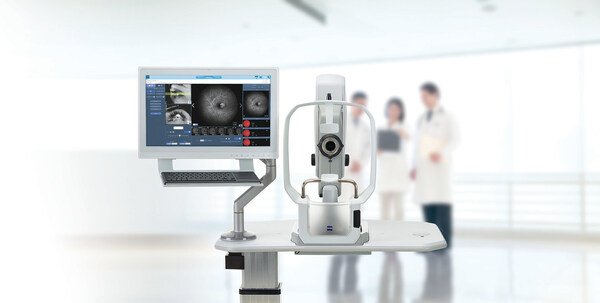
ZEISS CLARUS® 700 Receives NMPA Approval in China, Enhancing Retinal Diagnostics
ZEISS Medical Technology announced that the CLARUS® 700 from ZEISS has received National Medical Products Administration (NMPA) approval in China,...
June 06, 2025 | News
Innovent and HUTCHMED’s Sintilimab-Fruquintinib Combo Accepted for NMPA Review in Advanced Renal Cell Carcinoma
Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high quality me...
June 05, 2025 | News
Innovent Secures Second NMPA Breakthrough Therapy Designation for IBI363 in Advanced Lung Cancer
Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures, and commercializes high-quality m...
June 05, 2025 | News
JW Therapeutics Secures Macao Approval for CAR-T Therapy Carteyva®, Marking First Overseas Market Entry
JW Therapeutics (HKEx: 2126), an independent and innovative biotechnology company focusing on developing, manufacturing and commercializing cell immunother...
June 05, 2025 | News
Akeso’s Cadonilimab Secures NMPA Approval for First-Line Treatment of Cervical Cancer
Akeso, Inc. is pleased to announce that the National Medical Products Administration (NMPA) has approved the company's first- in-class PD-1/CTLA-4 bispecif...
June 05, 2025 | News
Singapore Approves Astellas’ VEOZA™ (Fezolinetant) for Moderate to Severe Menopausal Vasomotor Symptoms
Astellas Singapore has announced that the Health Sciences Authority (HSA) of Singapore has approved VEOZA™ (fezolinetant) 45 mg for the treatment of ...
June 04, 2025 | News
Mabwell Wins NMPA Approval for China’s First Albumin-Fused Long-Acting G-CSF Therapy MAILISHENG
Mabwell (688062.SH), an innovation-driven biopharmaceutical company with entire industry chain, announced that Albipagrastim alfa for Injection (trade name...
June 02, 2025 | News
Kexing Biopharm’s GB18 Receives IND Approval in China and U.S. for Cancer Cachexia Treatment
Kexing Biopharm (688136.SH) announced that the National Medical Products Administration (NMPA) has approved the Investigational New Drug (IND) applica...
May 30, 2025 | News
Adalvo Inks Licensing Deal with Formosa Pharmaceuticals for Novel Ophthalmic Steroid in Europe and Brazil
Adalvo announces the signing of an exclusive licensing agreement with Formosa Pharmaceuticals for the commercialisation of Clobetasol propio...
May 29, 2025 | News
Eisai’s DAYVIGO® Approved in China to Treat Insomnia, Targeting Over 170 Million Adults
Eisai Co., Ltd. announced that the in-house-discovered and developed orexin receptor antagonist DAYVIGO® (generic name: lemborexant) has been app...
May 29, 2025 | News
RemeGen’s Telitacicept Approved in China as First Dual-Target Biologic for Generalized Myasthenia Gravis
RemeGen Co., Ltd.(688331.SH / 09995.HK) announced that Telitacicept has officially been approved for marketing in China by the National Medical Produc...
May 28, 2025 | News
AnnJi Reports Promising Phase 1/2a Results for AJ201 in Rare Neuromuscular Disease SBMA
AnnJi Pharmaceutical Co., Ltd., a clinical-stage Taiwanese biotechnology company focused on addressing unmet medical needs in dermatology, neurology, and r...
May 22, 2025 | News
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks