BioPharma Drug Approval
BioAge Advances BGE-102 into Phase 1 Trials Triggering Milestone Payment to HitGen as DEL Technology Accelerates Drug Discovery
BGE-102, an orally available, brain-penetrant NLRP3 inhibitor, was developed from a hit compound identified using HitGen's industry-leading DEL technolog...
September 16, 2025 | News
Alphamab Oncology’s NDA for KN026 Accepted by NMPA for HER2-Positive Gastric Cancer
Alphamab Oncology announced that the New Drug Application (NDA) for anbenitamab injection (KN026), independently developed by the Company and co-developed ...
September 15, 2025 | News
China Grants First Approval for Tzield to Delay Onset of Type 1 Diabetes
Approval based on the TN-10 study, demonstrating Tzield’s ability to delay the onset of stage 3 T1D in adult and pediatric patients aged eight year...
September 11, 2025 | News
Takeda Wins U.S. FDA Approval to Expand VONVENDI Use in Adults and Children with Von Willebrand Disease
Approval Expands Use of VONVENDI to Include Routine Prophylaxis to Reduce the Frequency of Bleeding Episodes in Adults with Type 1 and 2 Von Willebrand D...
September 08, 2025 | Regulatory
Stallergenes Greer and Nuance Pharma Enter Exclusive Partnership to Develop and Commercialise Actair® in China
Stallergenes Greer, a global leader in allergy therapeutics, and Nuance Pharma, an innovation-focused biopharmaceutical company based in China, announced t...
September 05, 2025 | News
China’s Bon Natural Life Unveils Tea-Derived Compounds for Weight Management Through AI-Powered Drug Discovery
Bon Natural Life Limited , a leading bio-ingredient solution provider in the natural, health and personal care industry, announced a groundbreaking identif...
September 03, 2025 | News
Daiichi Sankyo Europe Initiates Development of Oral Triple Combination Tablets to Enhance LDL-C Management
Daiichi Sankyo Europe announced the initiation of the development of new oral triple combination tablets in Europe containing bempedoic acid, ezetimibe, an...
September 02, 2025 | News
FDA Approves Subcutaneous Leqembi Iqlik, Ushering in a New Era of Alzheimer’s Treatment
U.S. Food and Drug Administration (FDA) announced its approval of a subcutaneous formulation of Leqembi (lecanemab), branded as Leqembi Iql...
September 01, 2025 | News
China’s D3 Bio Secures FDA Breakthrough and Orphan Drug Designations for KRAS G12C Inhibitor D3S-001
Breakthrough Therapy Designation was granted for the treatment of adult patients with KRAS G12C-mutated locally advanced or metastatic non-small cel...
August 29, 2025 | News
MS Pharma Inaugurates Middle East’s First SFDA-Approved Biologics Manufacturing Facility In Saudi Arabia
MS Pharma inaugurates Middle East's first biologics manufacturing facility in Saudi Arabia, SFDA-approved and built to meet EMA and F...
August 28, 2025 | News
Abbott Secures Thailand Approval For Denosumab Biosimilar Expanding Access To Osteoporosis Treatment
Abbott denosumab biosimilar has received regulatory approval in Thailand, expanding access to advanced therapies for osteoporosis and cancer-related bone l...
August 27, 2025 | News
BioDlink Secures Indonesian Approval for Bevacizumab, Accelerating ASEAN and Global Expansion
BioDlink announced that its Bevacizumab Injection has obtained marketing authorization from Indonesia's National Agency of Drug and Food Con...
August 22, 2025 | News
Everest Medicines’ Etrasimod (VELSIPITY®) Receives Strong Recommendation in Updated ACG Clinical Guideline for Ulcerative Colitis
Everest Medicines (HKEX 1952.HK, "Everest", or the "Company"), a biopharmaceutical company focused on the discovery, clinical development, manufacturing, a...
August 18, 2025 | News
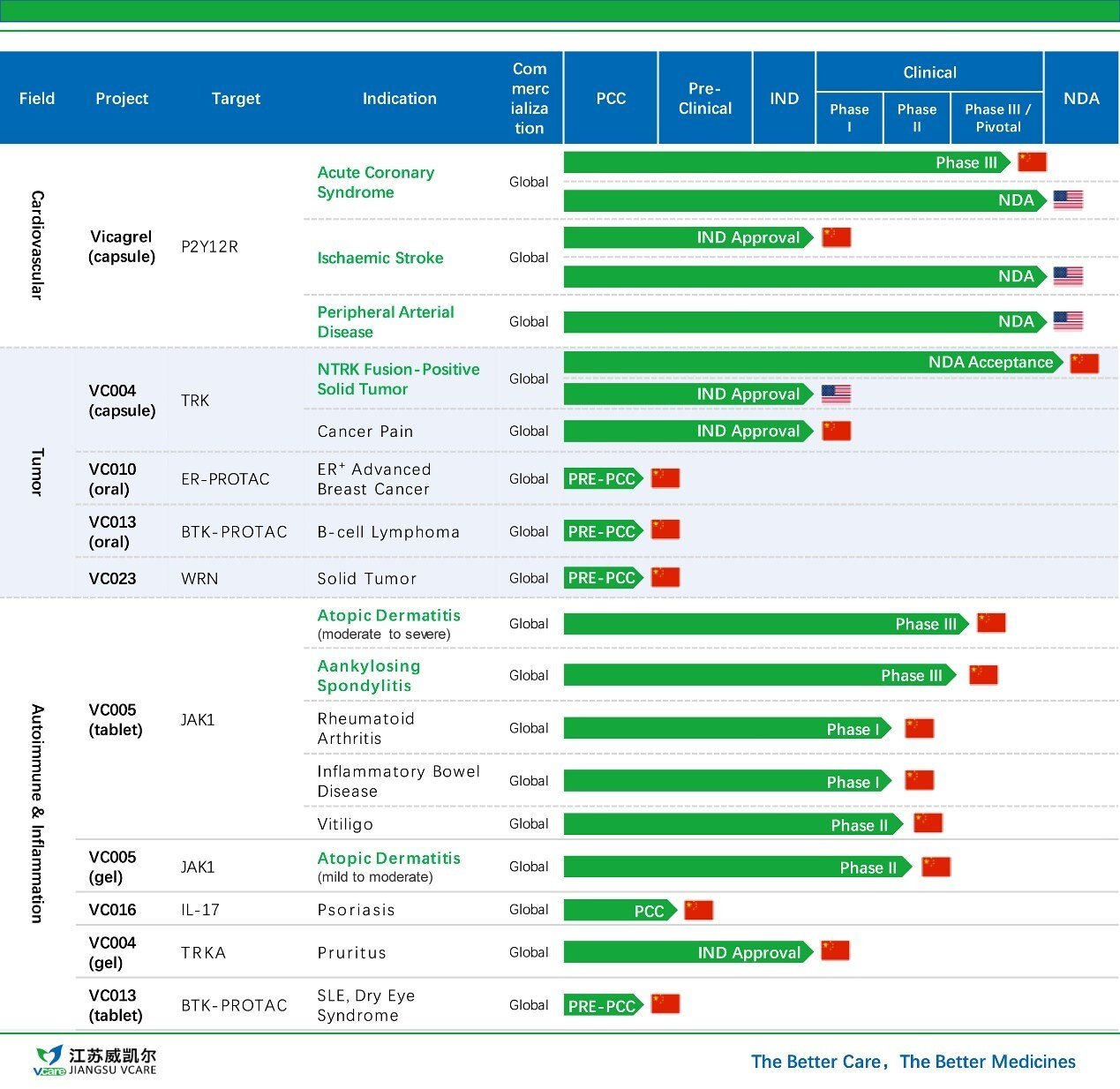
Jiangsu Vcare and Huadong Medicine Form Exclusive Partnership for Commercialization of VC005 Tablets in China
Jiangsu Vcare Pharmatech Co., Ltd. (Jiangsu Vcare) announced that it has entered into an exclusive strategic partnership with Huadong Medicine (H...
August 18, 2025 | News
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks